|
6–11 To overcome these problems, the primary task is to better understand the electrochemical behaviors that cover electronic/ionic transport properties and the charge transfer reaction at the electrode/electrolyte interface, since they govern the rate capability and cyclic stability, and would undoubtedly facilitate further electrode optimization.Įlectrochemical impedance spectroscopy (EIS) is one of the most powerful tools for the analysis of electrochemical processes that occur at electrode/electrolyte interfaces, 12 and has been widely used to study the electrochemical lithium intercalation in carbonaceous materials, 13,14 layer materials, 15–17 transition metal oxides, 18,19 special reaction mechanism materials 20,21 and sulfur cathodes. Typical weaknesses include poor cycle stability, unimpressive rate performance and unsatisfactory energy production due to the dissolved polysulfide intermediates (Li 2S x, 4 ≤ x ≤ 8) in the electrolyte, and the low electrical and ionic conductivity of elemental sulfur and its final discharge product. 1–5 Nevertheless, performances of Li–S batteries are not satisfactory as in theory. Lithium–sulfur (Li–S) batteries have become a hot topic due to their excellent specific capacity (1675 mA h g −1) and theoretical energy density (2600 W h kg −1). Introduction With the rapid development of electronic vehicles and large-scale power transfer installations, the demand for sustainable batteries with high energy density is increasing. Several important electrochemical reactions also have been verified by cyclic voltammetry curves. The inclined line arises from the diffusion process in the detectable potentials and frequency range. Based on detailed analysis of the change in kinetic parameters obtained from simulating the experimental EIS data as functions of potential, the high-frequency, middle-frequency and low-frequency semicircles/arcs can be attributed to the Schottky contact reflecting the electronic properties of materials, the charge transfer step and the formation of Li 2S respectively. 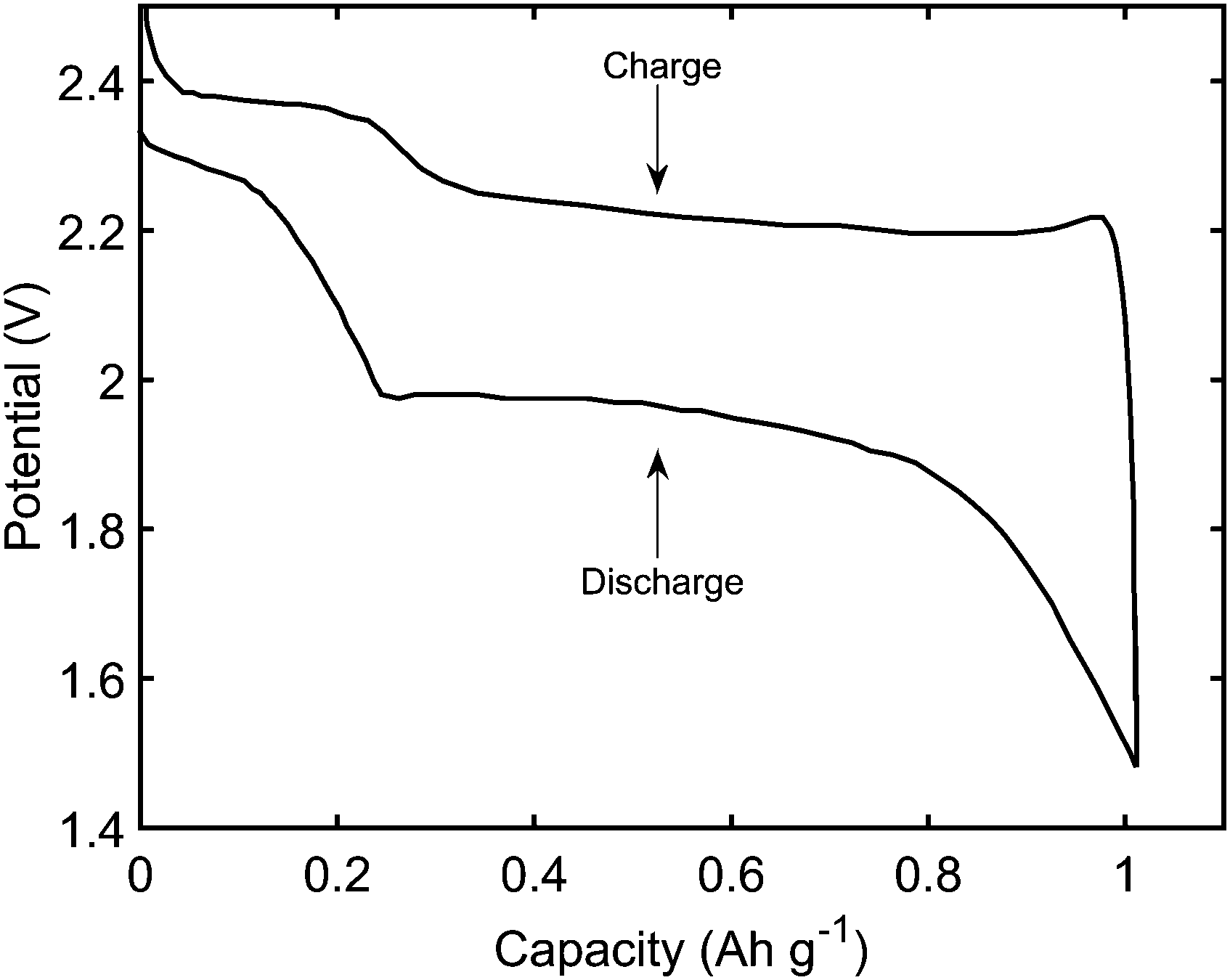
An appropriate equivalent circuit is proposed to fit the experimental EIS data. In the second discharge region (2.00–1.70 V), where soluble polysulfides are reduced to Li 2S, the EIS spectra exhibit three semicircles/arcs as the frequency decreased. Yang Q, Deng N, Zhao Y, Gao L, Cheng B, Kang W (2023) Chem Eng J 451:38532.Electrochemical impedance spectroscopy (EIS) was used to study the initial discharge/charge process in a sulfur cathode with different potentials. Umeshbabu E, Zheng B, Yang Y (2019) Recent progress in all-solid-state lithium−sulfur batteries using high Li-ion conductive solid electrolytes. Yang T, Qian T, Liu J, Xu N, Li Y, Grundish N, Yan C, Goodenough JB (2019) ACS Nano 13:9067–9073 Wang ZY, Wang L, Liu S, Li GR, Gao XP (2019). Zhang K, Chen Z, Ning R, Xi S, Tang W, Du Y, Liu C, Ren Z, Chi X, Bai M, Shen C, Li X, Wang X, Zhao X, Leng K, Pennycook SJ, Li H, Xu H, Loh KP et al (2019) ACS Appl Mater Interfaces 11:25147–25154 Zhou W, Chen H, Yu Y, Wang D, Cui Z, Disalvo FJ, Abruña HD (2013) ACS Nano 7:8801–8808 Rauh RD, Shuker FS, Marston JM, Brummer SB (1977) J Inorg Nucl Chem 39:1761–1766 The results of this study show the potential of the all-solid-state Li–S battery using the Li +-doped Na compound as the solid electrolyte and strongly indicate that a suitable solid electrolyte for Li–S batteries can be developed from the existing Na compounds. 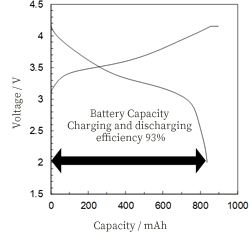
This is a pioneering study of the functioning of a Na-compound-based Li + conductor as a solid electrolyte for Li–S batteries. On the other hand, the discharge capacity was dropped to 380 mAh/g at 0.06 C, which could be due to insufficient electronic conduction in the composite and/or the homogeneity of the composite. This indicates that the overvoltage originates mainly from the IR drop of the solid electrolyte at a low rate. The discharge capacities did not change significantly when the discharge rate was increased to 0.03 C. For the cathode composite with sulfur, solid electrolyte, and carbon in a weight ratio of 2:7:1, an initial discharge capacity of 1480 mAh/g was obtained, and 760 mAh/g was maintained for the subsequent cycles at 0.006 C. The composition of the cathode, consisting of sulfur, solid electrolyte, and carbon, was optimized from the viewpoint of microstructure and charge–discharge performance. In this study, Li–S batteries were fabricated using 9(15NaI∙NaBH 4)∙LiI as the solid electrolyte, and their charge–discharge properties were investigated.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
